TODAY'S EDITION
April 26, 2024
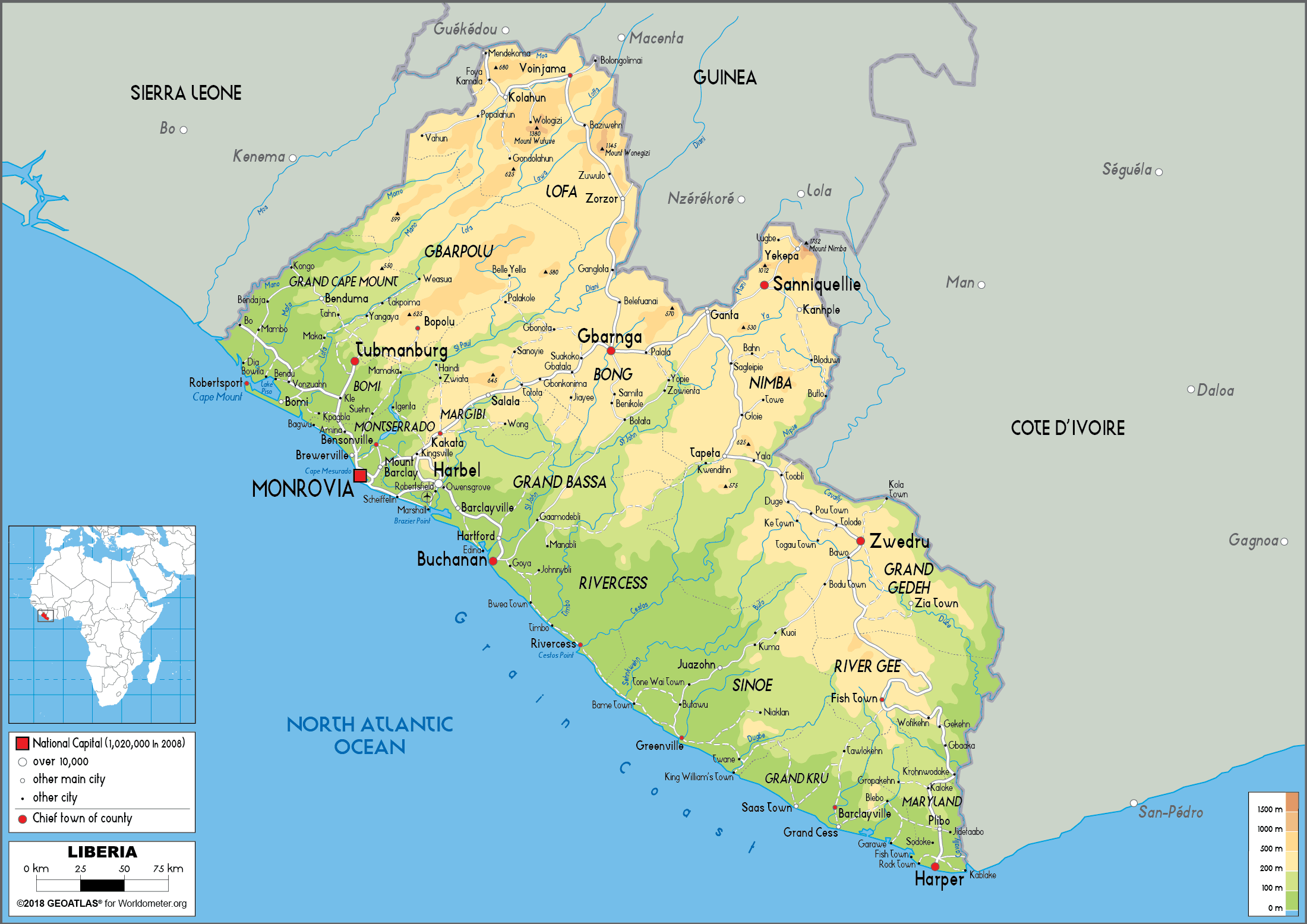
Senate probes alleged illicit mining in Gbarpolu
Two Gbarpolu County Senators have referenced a Chinese national named Gao Feng, who allegedly operates an illicit mining empire throughout Liberia, using…
April 26, 2024
Lawmakers in Dog fight
By Bridgett Milton Capitol Hill, Liberia, April 26, 2024—Drama erupts on the floor of the House of Representatives on Capitol…
April 26, 2024
Nimba loyalty shifting?
Should the National Elections Commission (NEC) confirm Nya Twayen’s win in Nimba by-election, discussions could now shift to who is…
April 26, 2024
Water crisis grips Feetuah
Residents of Feetuah town in Bong Country lack safe drinking water, and have resorting to fetching contaminated water from creeks…
April 26, 2024
GOL welcomes 2023 Human Rights Report on Liberia, but
By Kruah Thompson Monrovia, Liberia, April 26, 2024—The Government of Liberia welcomes the U.S. Department of State’s 2023 Human Rights…
April 26, 2024
Sen. McGill seeks US$1m for C.H. Rennie Hospital
By Ethel A. Tweh Margibi, Liberia, April 26, 2024 – Margibi County Senator Nathaniel F. McGill wants C.H. Rennie Hospital…